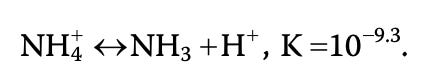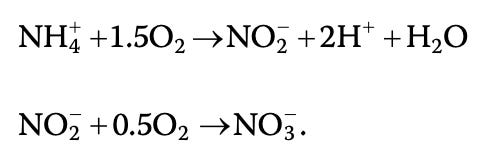Water | Part 2.2 - Dissolved Gases (Nitrogen Cycle)
Molecular nitrogen, ammonium, ammonia, nitrite, nitrate, and organic nitrogen
To analyze how water quality is monitored, understanding key components of water is an essential prerequisite. This series of newsletters will explore the key characteristics of both freshwater and seawater (e.g. solids, dissolved gas, temperature and pH), water balance in fish, and optimal physical and chemical ranges across species and production systems.
In our previous newsletters, we covered solids and DO in water. This week, let’s turn our attention to the nitrogen cycle.
Nitrogen Everywhere
As a key component of amino acids (i.e. units of protein), nitrogen is the building block of all organic life forms.
Nitrogen occurs in water in the following forms:
Molecular nitrogen (N2) - surface waters contain 10-15 mg/L dissolved molecular nitrogen (N2). However, molecular nitrogen is inert and only nitrogen fixing microorganisms can convert N2 into ammonium.
Ammonium (NH4+) - a form of nitrogen that is most easily consumed by microorganisms. Nitrogen incorporates into living cell via a process called assimilation.
Ammonia (NH3)
Nitrite (NO2-)
Nitrate (NO3-)
Organic nitrogen - digestible form of nitrogen for animals
On an aquaculture farm, nitrogen enters a system via nitrogen uptake through phytoplankton/plants or from manufactured feed pellets. Plants or phytoplanktons take in nitrate (NO3-) and ammonium (NH4+) and form essential amino acids for animal consumption. Farmed fish converts 20-40% of feed nitrogen into biomass, then secretes ammonia and urea as waste (via gills or as urine). Urea hydrolyzes in water, yielding ammonia and carbon dioxide. The unfinished feed and feces is decomposed into ammonia by microorganisms.
Ammonia
Formed from the metabolism of protein and as the major waste product of fish, total ammonia (measured as total ammonia-nitrogen TAN) exists in two forms: 1) ionized NH4+ (ammonium, non-toxic) and 2) un-ionized NH3 (ammonia, extremely toxic).
Ammonium and ammonia exist in a pH and temperature dependent equilibrium:
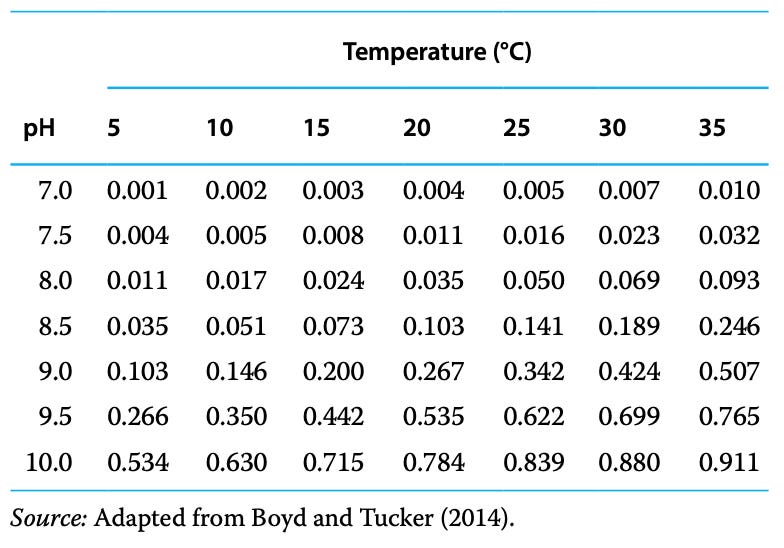
As pH and temperature increases, un-ionized NH3 increases relative to ionized NH4+. To calculate the % of un-ionized ammonia concentration in TAN, multiply total ammonia concentration by the percentage closest to the observed temperature and pH in water. For example, a total ammonia concentration of 5 ppm at pH 9 and 20 degrees Celcius would be = 5 ppm total ammonia x 26.7% = 1.335 ppm:
Key contributor to ammonia in water is fish excretion via gills. TAN concentration is regulated by rate of photosynthesis, decomposing farmed species and nitrifying bacteria in an aquaculture system:
Photosynthesis increases pH levels in water which favors a higher concentration of ammonia (the toxic form), thus ammonia concentration fluctuates with daily photosynthesis frequencies. During the day, pH is high and concentration of ammonia increases. At night, pH declines without photosynthesis. Ammonia concentration decreases in water, allowing for excretion through gills.
When plants, animals or microbes die, decomposition releases ammonia, minuscule particulate organic nitrogen (PON) and dissolved organic nitrogen (DON) into surrounding water. According to studies, organic matter with a C:N ratio of < 15 decomposes much faster and releases more ammonia.
Ammonia Toxicity
As ammonia concentration in water increases, ammonia excretion from fish reduces (due to decreased osmosis pressure) and levels of ammonia in blood and other tissues increases. Known as ammonia toxicity, elevated ammonia levels result in increased pH in blood and adverse effects on enzyme-catalyzed reactions and membrane stability.
Un-ionized ammonia begins causing gill damage at approximately 0.05 mg/L and death at approximately 2.0 mg/L.
The Fish Site - How to handle ammonia spikes when farming shrimp
Optimally, un-ionized ammonia concentrations in water should be < 0.5 mg/l. To prevent spike in NH3, water alkalinity should be maintained ≥ 40mg calcium carbonate per litre in neutral pH with high DO. Toxicity levels for un-ionized ammonia depend on the individual species; however, levels below 0.02 ppm are considered safe.
Detoxing - The Nitrogen Cycle
Un-ionized ammonia is oxidized to nitrate via nitrification in a two-step process carried out by nitrifying bacteria:
Step 1 - nitrosomonas bacteria decomposes ammonia (NH3) into nitrite (NO2-). The intermediate form of ammonia--nitrite--is known to be toxic to fish.
Step 2 - nitrite (NO2-) is converted to nitrate (NO3-) by nitrobacter bacteria. Nitrate can be utilized by plants. On some farms, denitrifying bacteria converts nitrate to nitrogen gas.
Abundance of nitrifying and denitrifying bacteria determines the balance of equation. High concentrations of nitrite occurs when the rate of ammonia oxidation to nitrite > rate of nitrite oxidation to nitrate. Nitrite also accumulates when denitrifying bacteria produce nitrite instead of nitrogen gas given specific conditions.
On some aquaculture farms, ammonia is removed from system using biofilter that includes substrate for both nitrosomonas bacteria and nitrobacter bacteria to grow.
Nitrite Poisoning
When fish absorb nitrite, ferrous iron in haemoglobin is oxidized to ferric iron and form methaemoglobin that prevents oxygen attachment. Significant amounts of blood carrying methaemoglobin causes the blood to turn brown, thus nitrite poisoning is known as the brown blood disease.
Tolerance to nitrite varies among species. Carp, catfish, tilapia, trout and salmon easily accumulate nitrite in bloodstream via the same gill anion-uptake mechanisms responsible for chloride uptake. Other species, for example temperate basses (Moronidae), exclude nitrite from bloodstream thus maintain relatively small amounts of nitrite in blood. Marine fish are generally tolerant of nitrite given high chloride concentration in seawater.
Given that NO(2)(-) and Cl(-) ions compete for the same active transport site, chloride concentrations can potentially reduce nitrite toxicity. To reduce nitrite concentration in water, add NaCI salt to increase the ratio of chloride to nitrite. A CI:NO2 ratio of 20:1 prevents negative side effects of high nitrite. Frequent water exchange and replacement also effectively reduces high levels of nitrite.
For more details on nitrogen, please refer to the following paper:
References
The Fish Site - Water quality: a priority for successful aquaculture
The Fish Site - How to achieve good water quality management in aquaculture
The Fish Site - A Fish Farmer's Guide to Understanding Water Quality - Part 1
The Fish Site - A fish farmer's guide to understanding water quality - Part 2
Global Seafood Alliance - Nitrogen a limiting factor in aquaculture production
Lucas John S et al. Aquaculture : Farming Aquatic Animals and Plants. Third ed. Wiley Blackwell 2019