Water quality monitoring is a priority for successful aquaculture. During a farm’s production cycle, disease-free water with optimal nutrient and air composition is key to maintaining a healthy stock. This is essential, as water quality deteriorates rapidly on a fish farm.
To analyze how water quality is monitored, understanding key components of water is an essential prerequisite. This series of newsletters will first explore the key characteristics of both freshwater and seawater, for example, solids, dissolved gas, temperature and pH. Followed with an understanding of water balance in fish and analysis on optimal physical and chemical factor ranges across species and production systems.
For farm managers and investors, it’s key to understand these metrics and ranges to determine the health of a farm and its subsequent production output.
What’s In Water - Part 1: Solids
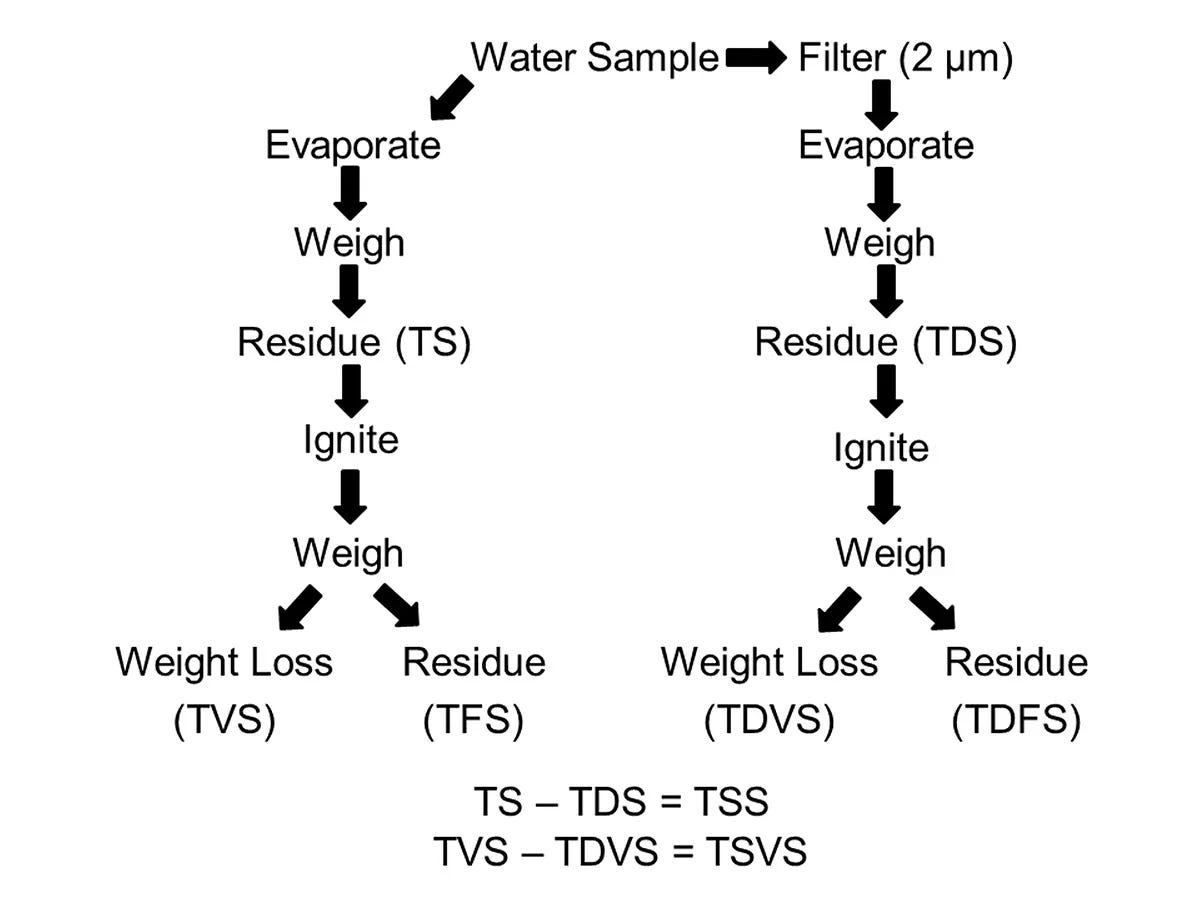
Solids are defined as the residue that remains after evaporation. It may be dissolved or suspended, and either inorganic or organic.
Solid Concentration
There are a few ways to measure solid concentration in water:
Total solids concentration (mg/L or ppm) = weight of residue remaining after an unfiltered water sample evaporates completely. Total solids comprise of total dissolved solids and total suspended solids.
Total dissolved solids (TDS) measure the weight of residue after complete evaporation of filtered water (with 2-μm apertures). TDS consists primarily of inorganic ions, including major cations (calcium Ca2+, magnesium Mg2+, sodium Na+ and potassium K+) and major anions (sulfate SO42-, chloride Cl– and bicarbonate/carbonate HCO3–/CO32-). Freshwater usually has a total dissolved solids concentration of <1000 mg/L. Seawater is especially high in sodium and chloride.
Total suspended solids consist of soil particles, phytoplankton, zooplankton and organic detritus (e.g. fish wastes and uneaten fish feeds). Clay particles are often suspended due to negative electrical charges.
Total volatile solids (TVS) = the weight loss following ignition at 500 °C of the total solid residue.
Particulate organic matter (POM) = ignition loss from the residue on the filter used for total suspended solids analysis. Elevated concentrations of POM are common in water bodies with moderate or high nitrogen and phosphorus inputs and an abundance of plankton
Depending on the concentration, dissolved solids (i.e. ions) may increase salinity of water and suspended solids may obscure the pathway of light in water.
Salinity
Expressed in parts per thousand (1‰ = 1000 mg/L), salinity is the total concentration of dissolved ions in water.
Aside from the major cations (calcium, magnesium, sodium and potassium) and anions (chloride, sulfate, bicarbonate, and carbonate), trace elements (iron, manganese, zinc, copper, boron, cobalt, molybdenum, ammonium, ammonia, nitrate, nitrite, phosphate) also play an important role in water. Lower pH allows for higher solubility of trace metals thus freshwater has higher trace metal concentration than seawater.
Total dissolved solids and salinity are of similar concentration in most waters, although salinity is more commonly used in discussions of marine waters. The average salinity of seawater is 34.5‰, while river water is usually less than 0.5‰.
Known as conductance, the ability of water to convey an electrical current increases with increasing ionic concentration. Hence, conductance can serve as an index to the degree of mineralization in water. Rainwater usually has a specific conductance of 10–20 μS/cm or less, while seawater conductance ranges around 50,000 μS/ cm.
For additional information on conductivity, proceed to Global Seafood Alliance’s two part series: Part 1 and Part 2.
Turbidity
Particles of low density and small sizes tend to stay suspended in water and settle slowly. Suspended solids often include plankton, fish wastes, uneaten fish feeds, or clay particles. As a rule, one pound of fish waste is produced for every pound of fish produced.
According to Stoke’s law, major factors impacting settling rate includes particle diameter, particle density and water temperature. Though not an obvious factor, as water temperature increases, the specific gravity and viscosity of water decreases resulting in faster settlement. Turbulence in water maintains particle suspension.
Unlike dissolved inorganic solids, dissolved organic solids (known as humic acids) color water and suspended solids reduce light penetration. Farms with high concentrations of dissolved organic substances and suspended particles tend to have lower rates of phytoplankton photosynthesis (i.e. less oxygen and nutrient generation). Turbidity levels > 20,000 ppm can alter fish behavior.
How to reduce turbidity?
By passing turbid water sources through a sedimentation basin before it enters a farming system, the amount of suspended particles in water decreases. In pond systems, growing plants to cover banks, lining embankments with plastic sheets and positioning aerators to minimize erosion are all effective methods to reduce turbidity.
Tiny Life in Water
Ranging from 0.2 μm to 20,000 μm, tiny phytoplankton (microscopic plants) and zooplanktons (microscopic animals) form the base of the food chain and can be categorized as both soluble particles (<2 μm) and insoluble particles. Phytoplankton produces large amounts of oxygen via photosynthesis and removes several forms of nitrogenous wastes, such as ammonia, nitrates, and urea.
Despite phytoplankton’s low densities, between 1.02 to 1.05 grams per cubic cm, they remain denser than water hence are subject to settling.
However, to remain in the upper layer of water for optimal light conditions, some planktons have evolved gas vacuoles to increase buoyancy and flagella for mobility. Organisms without evolved mobility ultimately depend on turbulence to remain within reach of light.
Phytoplankton is the main source of suspended organic matter in aquaculture ponds, measured via the concentration of chlorophyll A **(used in photosynthesis). Low phytoplankton density equates to less food and dissolved oxygen for fish. While too much phytoplankton can result in algal bloom with reduced light penetration and restricted oxygen supply that result in mass death.
References
The Fish Site - Water quality: a priority for successful aquaculture
The Fish Site - How to achieve good water quality management in aquaculture
The Fish Site - A Fish Farmer's Guide to Understanding Water Quality - Part 1
The Fish Site - A fish farmer's guide to understanding water quality - Part 2
Global Seafood Alliance - Dissolved and suspended solids in aquaculture systems
Lucas John S et al. Aquaculture : Farming Aquatic Animals and Plants. Third ed. Wiley Blackwell 2019